Unruptured Spinal Dermoid Cyst with Progressive Paraplegia
Wojak HJ, Henker C, Erbersdobler A and Piek J
DOI10.21767/2471-8041.100066
Wojak HJ1*, Henker C1, Erbersdobler A2 and Piek J1
1Department of Neurosurgery, University of Rostock, Rostock, Germany
2Department of Neurosurgery, and Pathology, University of Rostock, Rostock, Germany
- Corresponding Author:
- Wojak HJ
Department of Neurosurgery, University of Rostock, Rostock, Germany
Tel: +49 381 4980
E-mail: hansjoachim. wojak@med.uni-rostock.de
Received Date: July 04, 2017, Accepted Date: July 14, 2017, Published Date: July 16, 2017
Citation: Wojak HJ, Henker C, Erbersdobler A, Piek J (2017) Unruptured Spinal Dermoid Cyst with Progressive Paraplegia. Med Case Rep Vol.3 No.3: 31 doi:10.21767/2471-8041.100061
Abstract
Intramedullary dermoid cysts without spinal dysraphism are uncommon tumors. Only a few cases have been reported in the literature. We report a case of a 63-yearold woman with a spinal intramedullary dermoid cyst without rupture, but with progressive paraparesis and bladder dysfunction. Lumbar magnetic resonance imaging showed a large cystic lesion with inhomogeneous contrast enhancement and compression of the conus medullaris. Operation and histopathological examination confirmed a dermoid cyst. A follow up MRI imaging and examination was performed 4, 8 and 20 months after the operation. This case report adds to the few previous reported cases of intramedullary unruptured dermoid cysts and summarizes the latest literature.
Keywords
Dermoid cyst; Intramedullary tumor; Intramedullary cyst; Dermoid cyst without rupture
Introduction
Dermoid cysts are benign tumors which can occur in any localization. When located in the spine, they are usually found in the lumbar region and are often associated with congenital spinal dysraphism and/or dermal sinus tracts [1,2]. Arising from an embryologic miss-development during the ectodermal folds, dermoid cysts often present themselves in the 2nd or 3rd decade [3]. The symptoms result from their space-occupying effect causing irritation or compression of adjacent neural structures [3,4]. Especially the rupture of a dermoid cyst can be associated with a high morbidity and mortality [5].
Accounting for 0.7% to 1.8% of all tumors of the central nervous system, dermoid cysts are rarely found and described in the literature with only a few cases reported, especially in adults and intramedullary localization.
Case Report
A 63-year-old woman with therapeutic resistant lower back pain presented to our clinic. The pain started in summer 2012 without trauma and caused a progressive gait dysfunction. Since February 2013 the patient developed a mild but progressive paralysis in the left leg and a disturbance of passing water. Operative procedures or injections in the spine hadn’t been performed. Her further medical history included urinary tract infection, chronic renal insufficiency, non-insulin dependent diabetes mellitus type B, aortic ulcer, chronic heart disease, persistent oval foramen, adiposity, hypertension, nicotine abuse and hyperlipoproteinaemia. Cutaneous marks were absent.
Neurological examination of the cranial nerves and the upper extremities was normal. Strength was reduced in the limbs left>right (Hipflexion 3/5 left, 4/5 right; knee flexion and extension 4-/5 left, 4/5 right, ankle extension left 1/5, right 4/5, ancle flexion left 4/5, toe extension left 2/5. Sign of Laségue was positive at 10° on the left side. Deep tendon reflexes were symmetric. Pyramidal sign and myoclonus were negative. Standing and gait could not be judged due to the inability to stand upright. Urine could not be held back anal function was normal.
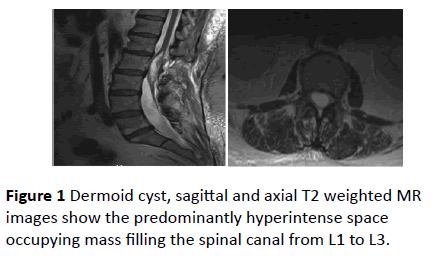
MRI of the lumbar spine showed a large cystic (50 × 19 × 15 mm) lesion with inhomogeneous contrast enhancement and compression of the conus medullaris on the left (Figures 1a and 1b). The patient was operated under the diagnosis of a cystic intramedullary, intradural tumor of the conus medullaris and tethered cord via laminectomy L1 and punction of the cyst.
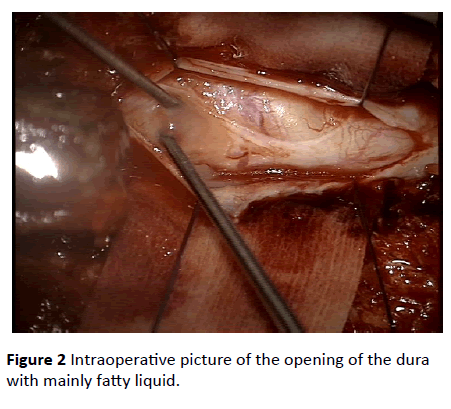
Intraoperatively (Figure 2), we found a distended dura which was dissected and the cyst was punctured after passing through a thick membrane of 2-3 mm. 3 ml of yellow doughy liquid could be evacuated and was sent for microbiological analysis. After opening the cyst with microdissectors, even more pus-like liquid was evacuated and purged. Altogether from the intraoperative aspect an intramedullary abscess was suspected.
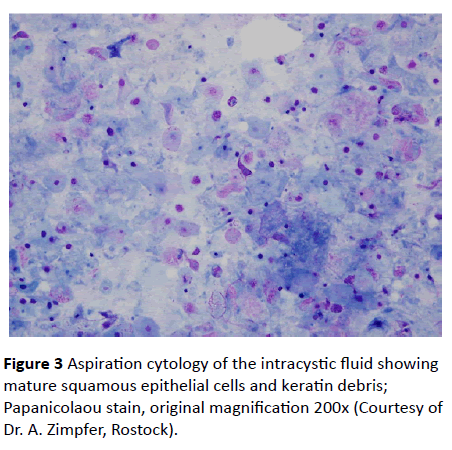
Thus, a calculated antibiotic therapy was initiated immediately after the operation. No bacteria however could be isolated from the intraoperatively evacuated liquid. Pathological examination revealed mature squamous epithelial cells and keratin debris, compatible with a dermoid cyst (Figure 3).
No atypical cells were found. There was no significant inflammation. Microorganisms were not detected. The postoperative course was uneventful with a continuous improvement during hospital and 3 weeks rehabilitation stay afterwards.
After 20 months, the patient was able to walk and climb stairs without limitation. The weakness improved continuously and only a slight paralysis of the left ankle extension could be seen.
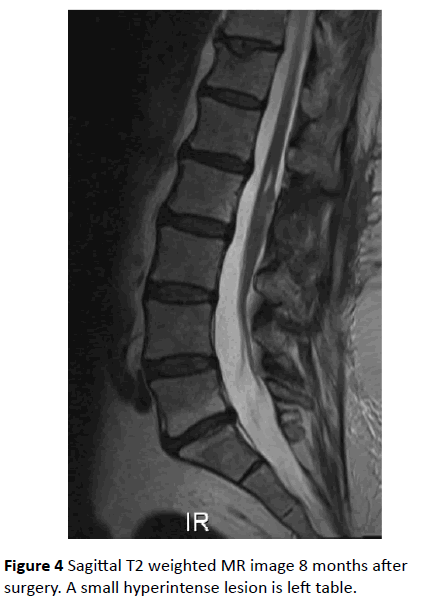
MR imaging 4, 8 and 20 months after the operation showed a good result with only a minor cyst left (Figure 4).
Discussion
Dermoid cysts are rare benign lesions within in the central nervous system and are usually located in the lumbosacral region [4,6]. Often combined with associated spinal dysraphism, their etiology is not completely understood. Congenital and acquired etiologies are discussed [7]. Due to the widely accepted theory of embryological malformation during the closure of the neural groove with ectoderm getting trapped under the lines of fusion of the ectodermal folds, dermoid cysts are not considered to be true neoplasms [8,9]. An extramedullary location (60%) is more common than an intramedullary one (40%) [4]. Lumbo-sacral accumulation may develop from the last closing, caudal part of the neural tube, making this region the most likely to be affected by cutaneous tissue getting trapped within the developing tube, giving rise to dermoid cyst [8]. Table 1 shows an overview of the previously reported cases of unruptured spinal dermoid cysts. Interestingly, many cases of children (<10 years of age) are presented.
| Authors | Number of Cases | Localization | Age of the patient (s) |
| Cervical/Thoracalnumbar | Child/Adult | ||
| Mishra SS, 2014 | 1 | t | c |
| Salo RK, 2013 | 1 | t | c |
| Futane SS, 2013 | 1 | I | c |
| Liu N, 2012 | 11 | I | a |
| Guidetti B, 1977 | 7 | I | a |
| Van Aalst J, 2009 | 4 | I | 3a/1c |
| Naoko K, 2007 | 1 | I | a |
| Kudo N, 2007 | 1 | I | a |
| Mhatre P, 2000 | 1 | I | a |
| Koplay M, 2015 | 1 | t | c |
| Guo S, 2014 | 5 | 2c/2t/1l | 3c/2a |
| Sanaullah M, 2013 | 1 | 1t/1 | c |
| Najjar MW, 2005 | 1 | t | a |
| ShikataJ, 1988 | 3 | 1t/21 | -- |
| ShubhaAM, 2010 | 5 | -- | c |
| Kwinta B, 2011 | 1 | I | a |
| Terai T, 2016 | 1 | I | a |
Table 1: Cases of unruptured spinal dermoid cysts in the literature (pubmed research) with their localization in the spine and the distinction between child or adult patients, “.” meaning no information available.
Imaging modality of choice is MR. The cysts present themselves as uni- or multilocular cystic lesions with varying intensity characteristics due to the variable cystic contents. The two major components, fluid and lipids, appear hypo- and hyperintense on T1-weighted MR images, respectively. According to the prevalent structure, the intensity may vary in different weighted images. Typically, enhancement of intravenous applicated contrast material is low, depending on the amount of cystic soft-tissue [9,10].
Various abnormalities occur with existing dermoid cysts, such as myelomeningocele, hypertrichosis, a dermal sinus tract, or a combination of these [3,9]. The histological distinction from epidermoid cysts arises from the dermal appendages like sebaceous glands, sweat glands, hair and hair follicles, being characteristic for dermoid cysts [9]. The thick yellow cystic material (Figure 3) typically contains desquamated epithelium and sebaceous gland secretion products [9].
Due to the slow growing character, symptoms occur usually not before the 2nd or 3rd decade of life [3] and arise from the compression of surrounding neural structures. All symptoms of a major space occupying lesion in the spinal cord can be seen, but vary in their intensity. A slight male predominance is described in the literature [3,4]. Although dermoid cysts are benign, two cases of malignant transformation have been described in the literature by Naoko in 2007.
Morbidity and mortality risks are associated with the intact cystic wall. In case of a rupture, a high degree of poor outcome is accompanied by lipid droplets scattered throughout the spinal cord [5]. While passive transportation of the lipid droplets via CSF pathway is easily understandable, the dissemination through the spinal cord into the intracranial cisterns and ventricles is thought to happen directly via the central canal, assuming an accompanying syringomyelic cavity which is normally rudimentary in adults [10].
A rupture, whether spontaneous, traumatic or intraoperative may remain asymptomatic or lead to headache, vomiting, nausea, meningism, or worsening of the neurological symptoms and can even result in coma. These symptoms are thought to arise from chemical or aseptic meningitis or arachnoiditis secondary to leakage of fat and proteinaceous material in the subarachnoid space [10].
Like in our case, subtotal resection is common in the treatment of intramedullary spinal cord tumors because the tumor capsule is frequently adhered tightly to the neural structures, leading to even more neurological deficits during forced total resection.
Conclusion
Here, in terms of differential diagnosis to epidermoid tumors, authors have questioned that due to the same clinical presentation, radiological examination and outcome, the histopathological differentiation between these tumors should be avoided and further named as ‘spinal cutaneous inclusion tumors’ [7]. As described in our case, the intraoperative differential diagnosis can be difficult and the tumor can be mistaken as a intramedullary abscess if further dermal appendages aren’t easily visible.
The strongest predicting factor of functional outcome is considered to be the preoperative neurological condition [11,12]. In our presented case, the patient did even improve after subtotal resection, leading to a good clinical result. MR follow up images show a good result of the cyst removal (Figure 4).
References
- Najjar MW, Kusske JA, Hasso AN (2005) Dorsal intramedullary dermoids. Neurosurg Rev 28: 320-325.
- Shubha AM, Mohanty S, Das K, Garg I (2010) Congenital inclusion tumors in the spinal dysraphism. Indian J Pediatr 77: 167-170.
- Cha JG, Paik SH, Park JS, Kim DH (2006) Ruptured spinal dermoid cyst with disseminated intracranial fat droplets. Br J Radiol 79: 167-169.
- Do-Dai DD, Brooks MK, Goldkamp A, Erbay S, Bhadelia RA (2010) Magnetic resonance imaging of intramedullary spinal cord lesions: A pictorial review. Curr Probl Diagn Radiol 39: 160-185.
- Altay H, KitiÃÆââ¬Â¦Ãâà ¸ O, Calli C, Yünten N (2006) A spinal dermoid tumor that ruptured into the subarachnoidal space and syrinx cavity. Diagn Interv Radiol 12: 171-173.
- Graham DV, Tampieri D, Villemure JG (1988) Intramdullary dermoid tumordiagnosed with the assistance of magnetic resonance imaging. Neurosurgery 23: 765-767.
- Van Aalst J, Hoekstra F, Beuls EA, Cornips EM, Weber JW, et al. (2009) Intraspinal dermoid and epidermoid tumors: report of 18 cases and reappraisal of the literature. Pediatr Neurosurg 45: 281-290.
- Patankar AP, Sheth JH (2012) Dermoid Cyst: A rare intramedullary inclusion cyst. Asian J Neurosurg 7: 81-83.
- De Maio PN, Mikulis DJ, Kiehl TR, Guha A (2012) Spinal conus dermoid cyst with lipid dissemination. Radiographics 32: 1215-1221.
- Calabrò F, Capellini C, Jinkins JR (2000) Rupture of spinal dermoid tumors with spread of fatty droplets in the cerebrospinal fluid pathways. Neuroradiology 42: 572-579.
- Sandalcioglu IE, Gasser T, Asgari S, Lazorisak A, Engelhorn T, et al. (2005) Functional outcome after surgical treatment of intramedullary spinal cord tumors : experience with 78 patients. Spinal Cord 43: 34-41.
- Kudo N, Hasegawa K, Ogose A, Hotta T, Watanabe K, et al. (2007) Malignant transformation of a lumbar intradural dermoid cyst. J Orthop Sci 12: 300-302.

Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences